A case study of non-surgical rhinoplasty to improve nasal deformity after septoplasty using Ellanse M dermal filler (polycaprolactone)
By Fabrizio Equizi
Abstract
A non-surgical solution to nasal deformity is becoming increasingly popular. This can be fillers to smooth out an minor bump on the bridge of the nose to fairly extensive augmentation to deformities acquired after surgery. Fillers can also be used to lift a droopy nose tip. Fillers in this area are not without risk. Strokes and blindness have been reported. A gradual approach building up small volumes of filler at intervals using a blunt cannula allows us to achieve impressive results with minimal risk and no downtime.
Introduction
This is a case study of a 42 year old male who was very keen to have treatment to improve the appearance of his nose which changed in shape after surgery 12 years ago.
Any treatment after surgery on the nose is more challenging because vascular supply to the nose is often compromised.
This study demonstrates a technique for re shaping the nose in a 3-step procedure using a novel, collagen stimulating filler made of polycaprolactone (Ellanse) and a blunt microcannula to minimise any risks.
The appearance of the nose was improved dramatically with no adverse effects apart from slight post procedure swelling.
Case Presentation
The patient had no issues with the shape and size of his nose until about 12 years ago after undergoing septoplasty or SMD (he couldn’t remember which) on the NHS for nasal obstruction. After the operation his nose became increasingly deformed to the point where it appeared squashed. He would often be asked if he’d been in an accident or a fight and his confidence was starting to be affected.
It was really obvious to him on photographs and prior to the initial consultation he emailed some of his own.
We discussed other options such as surgery but he was not keen on what that would entail (cartilage grafting) or what it may possibly cost. He was keen to pursue permanent fillers but I strongly advised against this treatment because of the higher risks of problems associated with permanent fillers, especially in a potential problem area like the nose.
Examination showed a Fitzpatrick 2 Glogau 2 male with an obvious saddle deformity of the nose associated with lateral indentations as a result of collapse of the nasal cartilage.
We discussed what fillers were available and in terms of the longevity he required we agreed on Ellanse M which should last for 2 years.
Of interest was the fact that his initial photos made the nose look much worse than mine did. Flash photography in this case whited out the shadows cast by the deformities, much like the way flash photography can make tear trough deformities appear less severe.
Diagnosis: post surgical saddle deformity of nose and collapse of nasal cartilage
Management and outcome
The management plan was to rebuild bridge of the nose and fill in the lateral indentations using a relatively new dermal filler called Ellanse which in the M variety should last 2 years.
Because of the previous surgery and the possible concern over vascular compromise it was agreed to build up the correction over 3 sessions using a cannula.
After photography in frontal, head down and 90 degree views the nose was covered in EMLA cream for topical anaesthesia. Nerve blockade was offered but he declined. Informed consent was obtained.
After half an hour the EMLA was wiped off and the nose cleaned with alcohol. A 25G needle was used to make a hole in the nasal tip for entry of a 27G 50mm cannula.
!ml of Ellanse M was mixed with 0.1 ml of mepivacaine 3% for added anaesthesia as Ellanse injection is associated with an after-sting for a few minutes.
Using a 50mm 27G cannula through the nasal tip it was possible to reach every part of the nose right up to the nasion. Using a few passes of the cannula the nasal bridge was built up by laying threads of filler along nose from the nasion, over the rhinion and down to the supratip.
Then the cannula was advanced laterally on each side to fill out any lateral indentation. In this first session 0.7mls was injected.
The procedure was repeated at 4 weeks then 10 weeks finishing off with little touch ups with a 27G needle. Subsequent top ups were 0.3ml and 0.1ml.
Even after the first session the degree of correction was impressive. This is clearly demonstrated on the appended photographs. The correction should be good for 2 years.
Patient was very pleased claiming that having the procedure had changed his life.
Discussion
Septoplasty is one of the most common procedures carried out in ENT surgery and can result in nasal deformities such as saddling and deviation. The same is also true after rhinoplasty. Incidence of aesthetic complications after septoplasty has been put at 1-8% and is thought to be due to excessive removal of or weakening if the septal cartilage structure.
Not everyone is keen on revision surgery due to cost or post-operative pain. Some deformities are very amenable to surgery. Han (2006) first described a “liquid” rhinoplasty using Restylane enriched with human fibroblasts.
The use of fillers in this area has to be done with caution. Necrosis and blindness have been reported after intravascular injections in nasal area. The vasculature of the nose has already been compromised after surgery, especially rhinoplasty, so too much filler under pressure may further compromise blood supply.
Ellanse is a filler composed of smooth bioresorbable microspheres (25-50μm) of polycaprolactone suspended in a carboxymethylcellulose carrier gel. In this respect it is very similar to Radiesse (which has spheres made of calcium hydroxylapatite)
Polycaprolactone has been used as a medical polymer since the 1980s with an excellent safety record (e.g. in absorbable sutures). It is broken down by hydrolysis into non toxic compounds like CO2 and water. The microspheres stimulate the formation of new collagen hence the longevity of the product. Depending on the length of the PCL chains this can be from 1-4 years.
In a similar fashion to Radiesse, Ellanse causes swelling after injection, hence the 3 step procedure. A post surgical nose is not the safest place to have any pressure from swelling or filler. The carrier gel gets absorbed before collagenesis can occur so top ups help maintain the cosmetic effect until neocollagenesis maintains the result.
The use of a blunt cannula minimises the risk of intravascular injection and most areas of the nose can be reached with a single entry point through the tip.

Patient self photograph

My baseline photograph

Pre Treatment

After 4 weeks

After final top up

After final top up

After final top up
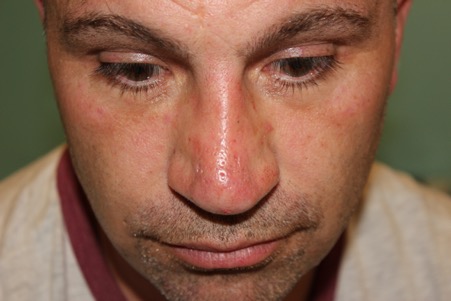
After second top up
Bray, D., C. Hopkins, and D. N. Roberts. “Injection Rhinoplasty: Non-Surgical Nasal Augmentation and Correction of Post-Rhinoplasty Contour Asymmetries with Hyaluronic Acid: How we do it.” Clinical Otolaryngology.3 (2010): 227. Print.
Han, Seung-Kyu, et al. “Augmentation Rhinoplasty using Injectable Tissue-Engineered Soft Tissue: A Pilot Study.” Annals of Plastic Surgery 56.3 (2006): 251-5. Print.
Iqbal, Kamran, Muhammad Ismail Khan, and Amir Amanullah. “Submucous Resection Versus Septoplasty: Complications and Functional Outcome in Adult Patients.” Gomal Journal of Medical Sciences 9.1 (2011): 23-7. Print.
Lazzeri, Davide, et al. “Blindness Following Cosmetic Injections of the Face.” Plastic and Reconstructive Surgery 129.4 (2012): 995-1012. Print.
Moers‐Carpi, Marion Michaela, and Sally Sherwood. “Polycaprolactone for the Correction of Nasolabial Folds: A 24‐Month, Prospective, Randomized, Controlled Clinical Trial.” Dermatologic Surgery (2013)Print.
Muhammad, I. A., and Rahman Nabil-ur. “Complications of the Surgery for Deviated Nasal Septum.” Journal of the College of Physicians and Surgeons–Pakistan : JCPSP 13.10 (2003): 565-8. Print.
Yeo, Nam-Kyung, and Yong Ju Jang. “Rhinoplasty to Correct Nasal Deformities in Postseptoplasty Patients.” American Journal Of Rhinology & Allergy 23.5 (2009): 540-5. Print.



